⏱️ 7 min read
Quantum physics stands as one of the most fascinating and mind-bending fields in modern science. It describes the behavior of matter and energy at the smallest scales—where particles act in ways that completely defy our everyday experience and common sense. While quantum mechanics has revolutionized technology and our understanding of the universe, it remains filled with phenomena that continue to surprise even seasoned physicists. Here are ten remarkable facts about the quantum world that showcase just how strange and wonderful reality becomes at the smallest scales.
Ten Fascinating Quantum Phenomena
1. Particles Can Exist in Multiple States Simultaneously
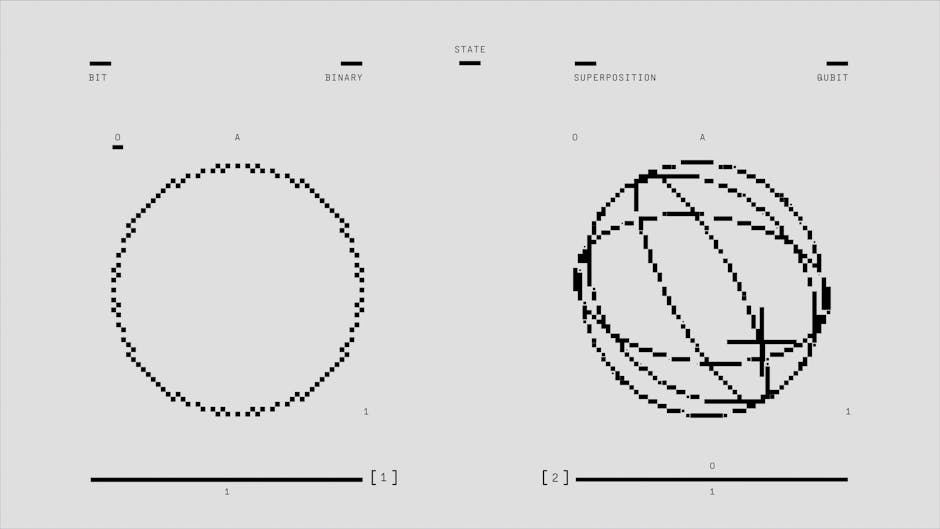
One of quantum physics' most counterintuitive principles is superposition—the ability of quantum particles to exist in multiple states at once until they're observed. An electron, for instance, doesn't have a definite position or spin direction until measured; instead, it exists in a probabilistic cloud of all possible states. This isn't just theoretical uncertainty—experiments have proven that particles genuinely occupy multiple states simultaneously. The famous Schrödinger's cat thought experiment illustrates this concept, suggesting a cat could theoretically be both alive and dead until observed. While macroscopic objects don't exhibit this behavior due to environmental interference, at the quantum level, superposition is fundamental to how particles behave.
2. Quantum Entanglement Creates Instantaneous Connections Across Space
When two particles become entangled, they form a connection that persists regardless of the distance separating them. Measuring the state of one entangled particle instantaneously determines the state of its partner, even if they're on opposite sides of the universe. Einstein famously called this "spooky action at a distance" because it seemed to violate the principle that nothing travels faster than light. However, numerous experiments have confirmed entanglement is real. In 2022, the Nobel Prize in Physics was awarded to scientists who proved that entanglement isn't due to hidden variables but represents a genuine quantum phenomenon. This strange connection is now being harnessed for quantum computing and ultra-secure communications.
3. You Cannot Know Everything About a Particle at Once
Heisenberg's Uncertainty Principle establishes fundamental limits on what we can know about quantum particles. Specifically, you cannot simultaneously measure both a particle's exact position and its exact momentum with perfect precision. The more accurately you determine one property, the less precisely you can know the other. This isn't a limitation of measurement technology—it's a fundamental feature of reality itself. The uncertainty principle arises from the wave-particle duality of quantum objects and has profound implications for everything from atomic structure to the existence of matter itself. It even explains why atoms don't collapse, as electrons can't be pinned down to exact positions within the nucleus.
4. Quantum Tunneling Allows Particles to Pass Through Barriers
In the quantum realm, particles can sometimes pass through barriers that should be impenetrable according to classical physics—a phenomenon called quantum tunneling. Because particles behave as probability waves, there's a small but non-zero chance they'll appear on the other side of an energy barrier they theoretically lack the energy to overcome. This isn't science fiction; tunneling is essential to many everyday technologies. The sun's fusion reactions depend on quantum tunneling, as do transistors in computer chips and tunnel diodes in electronics. Scanning tunneling microscopes use this effect to image individual atoms. Without quantum tunneling, stars wouldn't shine, and modern electronics wouldn't exist.
5. Empty Space Isn't Actually Empty
Quantum field theory reveals that what we call "empty space" is actually teeming with activity. Virtual particles constantly pop into and out of existence in pairs, borrowing energy from the vacuum for incredibly brief moments allowed by the uncertainty principle. These quantum fluctuations have measurable effects, such as the Casimir effect, where two metal plates placed extremely close together experience a small attractive force due to the quantum vacuum between them. This roiling quantum vacuum may even be connected to dark energy, the mysterious force accelerating the universe's expansion. The quantum vacuum represents one of physics' most profound concepts—that nothingness itself has structure and energy.
6. Observation Fundamentally Changes Quantum Systems
The act of measurement doesn't just reveal a quantum particle's properties—it actively changes them. This measurement problem lies at the heart of quantum mechanics' most puzzling aspects. The famous double-slit experiment demonstrates this perfectly: when electrons pass through two slits without being observed, they create an interference pattern characteristic of waves. But when detectors monitor which slit each electron passes through, the interference pattern disappears, and particles behave like classical objects. The mere act of observation collapses the quantum superposition, forcing particles to "choose" definite states. What qualifies as observation and why it has this effect remain subjects of ongoing debate among physicists and philosophers.
7. Quantum Computers Can Solve Certain Problems Exponentially Faster
While classical computers process information using bits that are either 0 or 1, quantum computers use quantum bits or "qubits" that can exist in superpositions of both states simultaneously. This allows quantum computers to perform certain calculations exponentially faster than classical machines. A quantum computer with just 300 qubits could theoretically perform more calculations simultaneously than there are atoms in the observable universe. Companies and research institutions have already built working quantum computers that have achieved "quantum supremacy"—solving specific problems impossible for classical computers. Though practical, large-scale quantum computing faces significant technical challenges, it promises to revolutionize fields from cryptography to drug discovery.
8. Particles and Waves Are Two Sides of the Same Coin
Wave-particle duality is the principle that all quantum objects exhibit both wave-like and particle-like properties, depending on how they're observed. Light, long understood as electromagnetic waves, also behaves as discrete particles called photons. Conversely, electrons and other particles that we typically think of as solid objects can produce interference patterns just like waves. This duality isn't that particles are sometimes waves and sometimes particles—rather, they're quantum objects that don't fit neatly into either classical category. The mathematics of quantum mechanics, particularly wave functions, elegantly describes this dual nature, representing quantum objects as probability waves that collapse into particle-like states upon measurement.
9. Quantum Physics Explains Why Matter Is Stable
The existence of stable atoms—and therefore all matter—depends entirely on quantum mechanics. Classical physics predicted that electrons orbiting atomic nuclei should continuously emit radiation and spiral into the nucleus within a fraction of a second, making stable atoms impossible. Quantum mechanics solved this crisis by showing that electrons occupy discrete energy levels rather than arbitrary orbits. Electrons can only exist in specific quantum states around a nucleus, and without external energy, they cannot fall to lower levels once they've reached the ground state. This quantization of energy levels, combined with the Pauli exclusion principle preventing multiple electrons from occupying identical states, explains the stability of atoms and the structure of the periodic table.
10. Quantum Mechanics Enables Teleportation of Information
Quantum teleportation—already achieved in laboratories—allows the transfer of quantum states between particles without physically moving the particle itself. Using entangled particles as a resource, scientists can transmit the complete quantum information of one particle to another distant particle, effectively "teleporting" its state. While this doesn't allow for Star Trek-style matter transmission (destroying and recreating objects elsewhere), it does enable the perfect transfer of quantum information. In 2017, Chinese scientists successfully teleported a photon's quantum state from Earth to a satellite 870 miles away. This technology is crucial for quantum communication networks and quantum internet, which promise unbreakable encryption and revolutionary computing capabilities.
The Quantum Revolution Continues
These ten facts barely scratch the surface of quantum physics' counterintuitive and fascinating nature. From superposition and entanglement to tunneling and wave-particle duality, quantum mechanics reveals a universe far stranger than our everyday experience suggests. What makes quantum physics particularly remarkable is that despite its weirdness, it's extraordinarily accurate—the most precisely tested theory in science. Technologies from lasers and semiconductors to MRI machines and atomic clocks all depend on quantum principles. As researchers develop quantum computers, quantum communication networks, and explore quantum gravity, the quantum revolution continues to reshape both our understanding of reality and the technologies that define modern life. The quantum world reminds us that the universe operates according to rules profoundly different from our intuitions—and that scientific exploration continues to reveal wonders we're only beginning to comprehend.